The overall goal of this project is to comprehend the formation of a key class of astrobiologically important molecules – alkylphosphonic acids – in interstellar ices as present in cold molecular clouds upon interaction with ionizing radiation. Cold molecular clouds are considered as nurseries of stars and planetary systems. The densest parts of these clouds eventually undergo gravitational collapse to form primitive material, which in turn supplies the basic ingredients for planets, their moons, and smaller bodies like asteroids and comets. Therefore, organic material such as alkylphosphonic acids, which initially formed in the molecular cloud, could have been incorporated into comets and parent bodies of meteorites such as of Murchison and might have been delivered to early Earth. Our project has unprecedented implications on multiple levels: to rationalize a potentially exogenous source of organic, water-soluble phosphorus-bearing compounds that might have initialized the organic phosphorus chemistry on early Earth, to better understand the origin of alkylphosphonic acids as detected in the Murchison meteorite, and to interpret prospective data from the Rosetta mission on the molecular composition of comet 67P/ Churyumov-Gerasimenko.

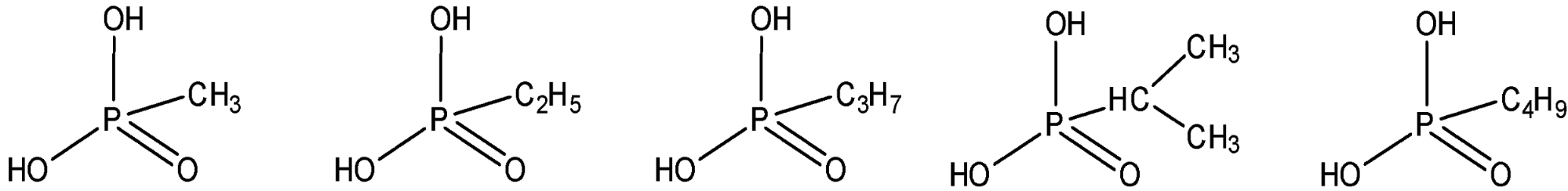

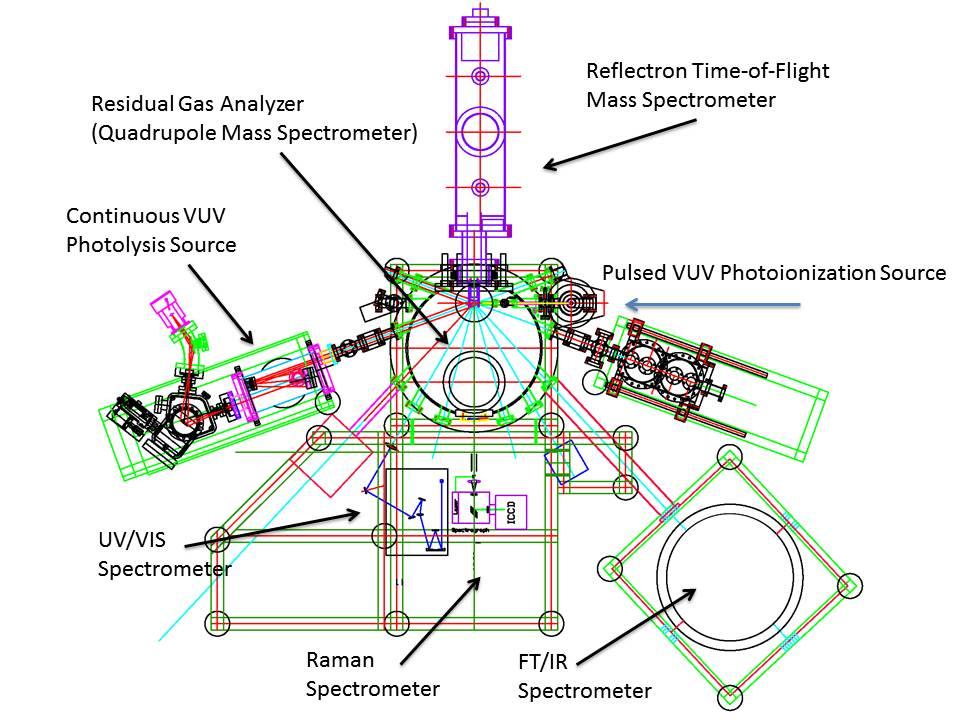
The methodology is based on conducting laboratory simulation experiments, which mimic the interaction of ionizing radiation with low temperature, astrophysically relevant ices, and tracing the formation of alkylphosphonic acids on line and in situ in the solid state and upon warm-up of the irradiated ices in the gas phase. We exploit novel, emerging laboratory techniques, which have not been accessible in previous simulation studies. These techniques are: photolysis of ices with monoenergetic photons (Ly-α 10.2 eV), a concerted on line and in situ infrared, Raman, and ultraviolet-visual (UVVIS) spectroscopy of the photolyzed ices, and - upon their release into the gas phase - fragment-free photoionization of alkylphosphonic acids with tunable vacuum ultraviolet (VUV) photons coupled with reflectron time-of-flight mass spectroscopy (ReTOF-MS). Organic residues, which could form upon annealing of the irradiated samples to room temperature, might also retain alkylphosphonic acids and hence are dissolved without hydrolyzing potential alkylphosphonic acids. The latter are derivatized and identified via gas chromatography – mass spectrometry (GC-MS) based on their known retention times and mass spectra. The GC-MS studies are carried out in collaboration with Cornelia Meinert (University of Nice, France).



Recent Selected Publications
1. A.M. Turner, M.J. Abplanalp, S.Y. Chen, Y.T. Chen, A.H.H. Chang, R.I. Kaiser A Photoionization Mass Spectroscopic Study on the Formation of Phosphanes in Low Temperature Phosphine Ices, Phys. Chem. Chem. Phys. 17, 27281-27291 (2015). (PDF)
2. A.M. Turner, M.J. Abplanalp, R.I. Kaiser, Probing the Carbon-phosphorus Bond Coupling in Low-temperature Phosphine (PH3)-Methane (CH4) Interstellar Ice Analogues, Ap. J. 819, 97 (2016). (PDF)
3. A.M. Turner, M.J. Abplanalp, T.J. Blair, R. Dayuha, R.I. Kaiser, An Infrared Spectroscopic Study Toward the Formation of Alkylphosphonic Acids and Their Precursors in Extraterrestrial Environments, ApJS, 234, 6 (2018). (PDF)
4. C. Zhu, R. Frigge, A.M. Turner, R.I. Kaiser, B-J Sun, S-Y Chen, A.H.H. Chang First identification of unstable phosphino formic acid (H2PCOOH), Chem. Comm. 54, 5716-5719 (2018). (PDF)
5. R. Frigge, C. Zhu, A.M. Turner, M.J. Abplanalp, B-J Sun, Y-S Huang, A.H.H. Chang, R.I. Kaiser, Synthesis of the hitherto elusive formylphosphine (HCOPH2) in the interstellar medium - a molecule with an exotic phosphorus peptide bond, Chem. Comm. 54, 10152-10155 (2018). (PDF)
6. A.M. Turner, A. Bergantini, M.J. Abplanalp, C. Zhu, S. Góbi, B-J Sun, K-H Chao, A.H.H. Chang, C. Meinert, R.I. Kaiser An interstellar synthesis of phosphorus oxoacids, Nature Comm. 9, 3851 (2018). (PDF)
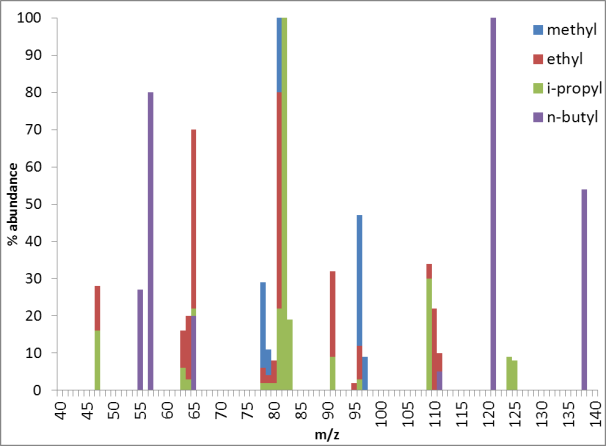
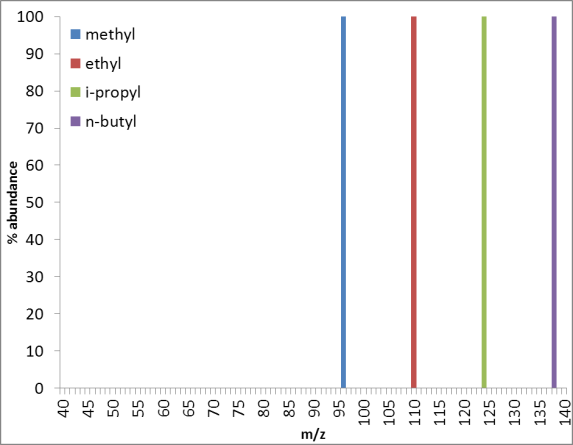
7. A. M. Turner, M. J. Aplanalp, A. Bergantini, R. Frigge, C. Zhu, B.-J. Sun, C.-T. Hsiao, A. H. H. Chang, C. Meinert, R.I. Kaiser, Origin of alkylphosphonic acids in the interstellar medium, Science Advances, 5, 8, eaaw4307 (2019). (PDF)
8. C. Zhu, A. K. Eckhardt, A. Bergantini, S. K. Singh, P. R. Schreiner, R. I. Kaiser, The elusive cyclotriphosphazene molecule and its Dewar benzene–type valence isomer (P3N3), Science Advances, 6, eaba6934 (2020). (PDF)
9. C. Zhu, A. M. Turner, M. J. Abplanalp, R. I. Kaiser, B. Webb, G. Siuzdak, R. C. Fortenberry, An Interstellar Synthesis of Glycerol Phosphates, Astrophys. J. Lett., 899, L3 (2020). (PDF)
10. C. Zhu, A. Bergantini, S. K. Singh, R. I. Kaiser, A. K. Eckhardt, P. R. Schreiner, Y.-S. Huang, B.-J. Sun, A. H. H. Chang, Formation of phosphine imide (HN=PH3) and its phosphinous amide (H2N–PH2) isomer, Chem. Comm., 57, 4958-4961 (2021). (PDF)


