
The primary objectives of this project are to experimentally explore the energetics, dynamics, and potential energy surfaces of elementary reactions of the simplest silicon-bearing diatomic radical, silylidyne (SiH), with prototype hydrocarbon molecules. The proposed studies explore the formation of small organosilicon molecules under single collision conditions as provided in crossed molecular beam experiments. To achieve these objectives, we investigate the energy-dependent chemical reaction dynamics of silylidyne radicals under single collision conditions in an ultra-clean crossed molecular beam machine. This setup incorporates a universal, rotatable angular resolved time-of-flight mass spectrometric detector with hard and soft electron impact ionization as well as laser induced fluorescence (LIF) detection.By conducting state-of-the-art crossed molecular beam experiments and combining these results with ab initio and quasi classical trajectory (QCT) calculations, we will extract the chemical dynamics, reaction mechanisms, products, intermediates, energetics, branching ratios, and enthalpies of formation of small organosilicon molecules over a broad range of collision energies from 2 to 45 kJmol-1. Besides the basic scientific interest from the physical chemistry community in unraveling the chemical dynamics of hitherto poorly explored silylidyne radical (SiH) reactions and comparing them to those of the isoelectronic methylidyne radical (CH), the proposed studies address fundamental questions of interest to the astrochemistry and physical-organic chemistry communities. This leads to a detailed understanding of the formation and role of small organosilicon molecules in the interstellar medium (astrochemistry) and advances insights into chemical bonding and unusual (cyclic) structures incorporating a ‘heavy’ main group element (silicon) (physical organic chemistry).
First, we commissioned and operated a hydrocarbon-free crossed beam machine with ultra-low background detection schemes incorporating a novel, pulsed supersonic beam source of ground state silylidyne radicals (SiH(X2Π)) generated via photolysis of helium-seeded disilane (Si2H6). The radical beam was exploited to untangle the previously elusive energetics and dynamics of silylidyne radical reactions with a homologues series of alkynes acetylene (C2H2), methylacetylene (CH3CCH), and dimethylacetylene (CH3CCCH3). The detection of 1-silacycloprop-2-enylidene (SiC2H2; (1)), 2-methyl-1-silacycloprop-2-enylidene (SiC2HCH3; (2)), and 2,3-dimethyl-1-silacycloprop-2-enylidene (SiC2(CH3)2; (3)) manifested the formation of three representatives of the hitherto obscure class of partially aromatic organosilicon molecules: silacyclopropenylidenes. In all systems, the silylidyne radical added without entrance barrier to the carbon-carbon triple bond yielding eventually cyclic collision complexes (indirect scattering dynamics). The latter decompose via loose exit transition states in overall exoergic reactions of up to 30 kJ mol-1 via hydrogen emission from the silylidyne moiety to silacyclopropenylidene molecules (1)-(3). Crossed beam reactions of silylidyne with allene (H2CCCH2) and 1,3-butadiene (H2CCHCHCH2) - structural isomers of methylacetylene and of dimethylacetylene - carrying cummulenic and conjugated carbon-carbon double bonds, respectively, revealed distinct reaction dynamics leading to the formation of 2-methyl-1-silacycloprop-2-enyidene (SiC2HCH3; (2)) and 1-silacyclopenta-2,4-diene (SiC4H6; (4)), respectively. The exploitation of D4-allene (D2CCCD2) provided evidence of two discrete reaction mechanisms involving atomic hydrogen and deuterium loss pathways. Second, we explored in crossed molecular beam reactions of silylidyne radicals with molecular oxygen (O2) and of electronically excited silicon atoms (Si(1D)) with allene (H2CCCH2) and methylacetylene (CH3CCH) the formation of key building blocks of interstellar silicates and of silicon carbide nanoparticles, respectively. We demonstrated that silicon dioxide (SiO2) along with silicon monoxide (SiO) is synthesized under single collision conditions providing a low-temperature path to the formation and growth of silicates in the interstellar medium.


Third, we expanded the investigation of the chemical dynamics of silicon-bearing molecules holding unusual chemical bonding by conducting crossed molecular beam reactions of ground state atomic carbon (C(3P)) and silicon (Si(3P)) with silane (SiH4) and disilane. In the silicon – silane system, the hitherto elusive disilavinylidene (H2SiSi) molecule, which is in equilibrium with the mono-bridged (Si(H)SiH) and di-bridged (Si(H2)Si) isomers, was formed. The reaction involves fascinating dynamics via an initial formation of a van der Waals complex in the entrance channel, a submerged barrier to insertion, intersystem crossing from the triplet to the singlet manifold, and hydrogen migrations. These studies provide a rare glimpse at the largely obscure silicon chemistry on the molecular level and shed light on the remarkable non-adiabatic reaction dynamics of silicon, which are quite distinct from those of isovalent carbon system (C(3Pj)–CH4), which shows no reaction under analogous experimental conditions. This provides important insight revealing an exotic silicon chemistry to disilavinylidene on the most fundamental, microscopic level. Non-adiabatic reaction dynamics via molecular hydrogen loss were also unraveled for the silicon – disilane system; the cyclic/hydrogen-bridged Si3H4 product(s) underline(s) extensive isomerization from the initial collision complex(es).
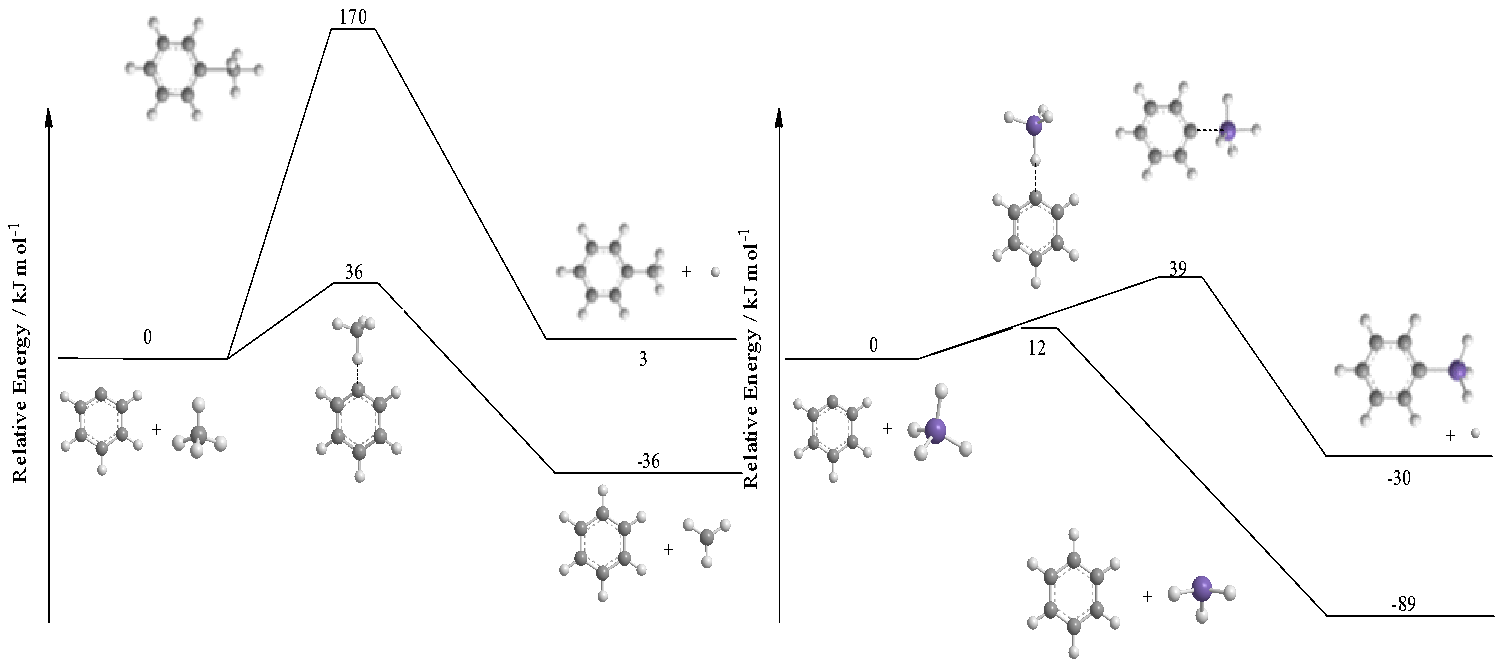
Finally, we investigated the reaction dynamics of the phenyl radical (C6H5; X2A1) with silane (SiH4) under single collision conditions revealing the formation of phenylsilane (C6H5SiH3) plus atomic hydrogen (H) via direct scattering dynamics through a transition state involving a simultaneous silicon-carbon bond formation and a silicon-hydrogen bond rupture via a trigonal bipyramidal transition state (Fig. 5). Compared to the isovalent methane (CH4) – phenyl radical (C6H5) system, the replacement of a single carbon atom by silicon lowers the barrier to substitution by about 130 kJmol-1 thus making the substitution pathway in the silane – phenyl system competitive with the hydrogen abstraction channel than in the methane – phenyl system, which only shows an abstraction to benzene (C6H6) plus methyl (CH3). Therefore, reactions with silicon-bearing species proposed here shed light on an unexplored silicon chemistry and hitherto unobserved chemical dynamics providing fundamental insights into novel reaction mechanisms via radical substitution pathways through exotic transition states.

Abstraction
Substitution
Recent Selected Publications
1. R.I. Kaiser, X. Gu, Chemical Dynamics of the Formation of the Ethynylsilylidyne Radical (SiCCH(X2Π)) in the Crossed Beam Reaction of Ground State Silicon Atoms (Si(3P)) with Acetylene (C2H2(X1Σg+)), JCP 131, 104311/1-6 (2009). (PDF)
2. D.S.N. Parker, A.W. Wilson, R.I. Kaiser, T. Labrador, A.M. Mebel, Synthesis of the Silaisocyanoacetylene Molecule, JACS, 134, 13896-13901 (2012). (PDF)
3. D.S.N. Parker, A.V. Wilson, R.I. Kaiser, T. Labrador, A.M. Mebel, Gas-Phase Synthesis of the Silaisocyanoethylene Molecule (C2H3NSi), JOC, 77, 8574 (2012). (PDF)
4. R.I. Kaiser, S.P. Krishtal, A.M. Mebel, O. Kostko, M. Ahmed, An Experimental and Theoretical Study of the Ionization Energies of SiC2Hx (x = 0, 1, 2) Isomers. Ap. J., 761, 178 (2012). (PDF)
5. D.S.N. Parker, A.V. Wilson, R.I. Kaiser, N.J. Mayhall, M. Head-Gordon, A.G.G.M. Tielens, On the Formation of Silacyclopropenylidene (c-SiC2H2) and its Role in the Organosilicon Chemistry in the Interstellar Medium, Ap. J., 770, 33 (2013). (PDF)
6. B. B. Dangi, Y.S. Kim, S.A. Krasnokutski, R.I. Kaiser, C.W. Bauschlicher Jr, Toward the Formation of Carbonaceous Refractory Matter in High Temperature Hydrocarbon-rich Atmospheres of Exoplanets Upon Micrometeoroid Impact, Ap. J., 805:76, 1-7 (2015). (PDF)
7. S. Thorwirth, R.I. Kaiser, K.N. Crabtree, M. C. McCarthy, Spectroscopic and structural characterization of three silaisocyanides: exploring an elusive class of reactive molecules at high resolution, Chem. Comm. 51, 11305 (2015). (PDF)
8. T. Yang, B.B. Dangi, P. Maksyutenko, R.I. Kaiser, L.W. Bertels, and M. Head-Gordon, Combined Experimental and Theoretical Study on the Formation of the Elusive 2-Methyl-1-silacycloprop-2-enylidene Molecule under Single Collision Conditions via Reactions of the Silylidyne Radical (SiH; X2Π) with Allene (H2CCCH2; X1A1) and D4-Allene (D2CCCD2; X1A1), J. Phys. Chem. A, 119, 12562-12578 (2015). (PDF)
9. T. Yang, B.B. Dangi, A.M. Thomas, R.I. Kaiser, Untangling the Reaction Dynamics of the Silylidyne Radical (SiH; X2Π) with Acetylene (C2H2; X1Σg+), Chem. Phys. Lett., 654, 58-62 (2016). (PDF)
10. T. Yang, B.B. Dangi, A.M. Thomas, B-J Sun, T-J Chou, A.H.H. Chang, R.I. Kaiser, Gas-Phase Synthesis of 1-Silacyclopenta-2,4-diene, Angew. Chem. Int. Ed, 55, 7983-7987 (2016). (PDF)
11. T. Yang, B.B. Dangi, R.I. Kaiser, L.W. Bertels, M. Head-Gordon, A Combined Experimental and Theoretical Study on the Formation of the 2-Methyl-1-silacycloprop-2-enylidene Molecule via Crossed Beam Reactions of the Silylidyne Radical (SiH; X2Π) with Methylacetylene (CH3CCH; X1A1) and D4-Methylacetylene(CD3CCD; X1A1), J. Phys. Chem. A, 120, 4872-4883 (2016). (PDF)
12. G. Tarczay, M. Förstel, P. Maksyutenko, R.I. Kaiser, Formation of Higher Silanes in Low-Temperature Silane (SiH4) ices, Inorg. Chem., 55, 8776-8785 (2016). (PDF)
13. T. Yang, A.M. Thomas, B.B. Dangi, R.I. Kaiser, M-H Wu, B-J Sun, A.H.H. Chang, Formation of the 2,3-Dimethyl-1-silacycloprop-2-enylidene Molecule via the Crossed Beam Reaction of the Silylidyne Radical (SiH; X2Π) with Dimethylacetylene (CH3CCH3; X1A1g), J. Phys. Chem. A, 120, 7262-7268 (2016). (PDF)
14. T. Yang, B.B. Dangi, A.M. Thomas, R.I. Kaiser, B-J Sun, M. Stas, A.H.H. Chang, Gas-Phase Synthesis of the Elusive Trisilicontetrahydride Species (Si3H4), J. Phys. Chem. Lett., 8, 131-136 (2017). (PDF)
15. T. Yang, B.B. Dangi, R.I. Kaiser, K-H Chao, B-J Sun, A.H.H. Chang, T.L. Nguyen, J.F. Stanton, Gas-Phase Formation of the Disilavinylidene (H2SiSi) Transient, Angew. Chem. Int. Ed., 56, 1264-1268 (2017). (PDF)
16. G. Tarczay, M. Förstel, S. Góbi, P. Maksyutenko, R.I. Kaiser, Synthesis of the Smallest Member of the Silylketene Family: H3SiC(H)CO, Chem. Phys. Chem. 18, 882-889 (2017). (PDF)
17. T. Yang, A.M. Thomas, B.B. Dangi, R.I. Kaiser, A.M. Mebel, T.J. Millar, Directed Gas Phase Formation of Silicon Dioxide and Implications for the Formation of Interstellar Silicates, Nat. Commun., 9, 774 (2018). (PDF)
18. A.M. Thomas, B.B. Dangi, T. Yang, R.I. Kaiser, L. Lin, T-J Chou, A.H.H. Chang Are Nonadiabatic Reaction Dynamics the Key to Novel Organosilicon Molecules? The Silicon (Si(3P))-Dimethylacetylene (C4H6(X1A1g)) System as a Case Study, J. Phys. Chem. Lett. 9, 3340-3347 (2018). (PDF)
19. M. Lucas, A. M. Thomas, T. Yang, R. I. Kaiser, A. M. Mebel, D. Hait, and M. Head-Gordon, Bimolecular Reaction Dynamics in the Phenyl-Silane System: Exploring the Prototype of a Radical Subsitition Mechanism, J. Phys. Chem. Lett. 9, 5135-5142 (2018). (PDF)
20. A.M. Thomas, B.B. Dangi, T. Yang, R.I. Kaiser, B.-J. Sun, T.-J. Chou, A.H.H. Chang, A Crossed Molecular Beams Investigation of the Reactions of Atomic Silicon (Si(3P)) with C4H6 Isomers (1,3-Butadiene, 1,2-Butadiene, and 1-Butyne), Chem. Phys. 520, 70-80, (2019). (PDF)
21. A. M. Thomas, B. B. Dangi, T. Yang, G. Tarczay, R. I. Kaiser, B.-J. Sun, S.-Y. Chen, A. H. H. Chang, T. L Nguyen, J. F. Stanton, A. M. Mebel, Directed Gas-Phase Formation of the Germaniumsilylene Butterfly Molecule (Ge(μ-H2)Si), J. Phys. Chem. Lett. 10, 1264−1271 (2019). (PDF)
22. T. Yang, L. Bertels, B.B. Dangi, X. Li, M. Head-Gordon, R.I. Kaiser, Gas phase formation of c-SiC3 molecules in the circumstellar envelope of carbon stars, Proceedings of the National Academy of Sciences, 116, 14471-14478 (2019). (PDF)
23. S. Doddipatla, C. He, R. I. Kaiser, Y. Luo, R. Sun, G. R. Galimova, A. M. Mebel, T. J. Millar, A Chemical Dynamics Study on the Gas Phase Formation of Thioformaldehyde (H2CS) and its Thiohydroxycarbene Isomer (HCSH), Proc. Natl. Acad. Sci. U.S.A., 117, 22712-22719, (2020). (PDF) (Supplemental Information)
24. Z. Yang, C. He, S. Doddipatla, V. S. Krasnoukhov, V. N. Azyazov, A. M. Mebel, R. I. Kaiser, Gas Phase Formation of Methylgermylene (HGeCH3), ChemPhysChem, 21, 1898-1904 (2020). (PDF)
25. S. Doddipatla, Z. Yang, A. M. Thomas, Y. Chen, B. Sun, A. H. H. Chang, A. M. Mebel, R. I. Kaiser, Gas Phase Synthesis of the Elusive Trisilacyclopropyl Radical (Si3H5) via Unimolecular Decomposition of Chemically Activated Doublet Trisilapropyl Radicals (Si3H7), J. Phys. Chem. Lett., 11, 7874-7881 (2020). (PDF) (Supplemental Information)
26. Z. Yang, S. Doddipatla, C. He, V. S. Krasnoukhov, V. N. Azyazov, A. M. Mebel, R. I. Kaiser, Directed Gas Phase Formation of Silene (H2SiCH2), Chem. Eur. J., 26, 13584-13589 (2020). (PDF)
27. Z. Yang, S. Doddipatla, R. I. Kaiser, V. S. Krasnoukhov, V. N. Azyazov, A. M. Mebel, Directed Gas Phase Formation of the Elusive Silylgermylidyne Radical (H3SiGe, X2A''), ChemPhysChem, 22, 184-191 (2021). (PDF)
28. Z. Yang, S. Doddipatla, R. I. Kaiser, A. A. Nikolayev, V. N. Azyazov, A. M. Mebel, On the Synthesis of the Astronomically Elusive 1-Ethynyl-3-Silacyclopropenylidene (c-SiC4H2) Molecule in Circumstellar Envelopes of Carbon-rich Asymptotic Giant Branch Stars and Its Potential Role in the Formation of the Silicon Tetracarbide Chain (SiC4), Astrophys. J. Lett., 908, L40 (2021). (PDF)
29. C. He, Z. Yang, S. Doddipatla, L. Zhao, S. Goettl, R. I. Kaiser, M. X. Silva, B. R. L. Galvão, Non-Adiabatic Reaction Dynamics in the Gas-Phase Formation of Phosphinidenesilylene, the Isovalent Counterpart of Hydrogen Isocyanide, under Single-Collision Conditions, J. Phys. Chem. Lett., 12, 2489-2495 (2021). (PDF)
30. Z. Yang, B.-J. Sun, C. He, S. Goettl, Y.-T. Lin, A. H. H. Chang, R. I. Kaiser, Combined Experimental and Computational Study on the Reaction Dynamics of the D1-Silylidyne (SiD) - Silane (SiH4) System, J. Phys. Chem. A., 125, 2472-2479 (2021). (PDF)
31. Z. Yang, C. He, S. Goettl, R. I. Kaiser, Reaction Dynamics Study of the Molecular Hydrogen Loss Channel in the Elementary Reactions of Ground State Silicon Atoms (Si(3P)) with 1- and 2-Methyl-1,3-Butadiene (C5H8), J. Phys. Chem. A., 125, 5040-5047 (2021). (PDF)
32. S. J. Goettl, S. Doddipatla, Z. Yang, C. He, R. I. Kaiser, M. X. Silva, B. R. L. Galvão, T. J. Millar, Chemical Dynamics Study on the Gas-Phase Reaction of the D1-Silylidyne Radical (SiD; X2Π) with Deuterium Sulfide (D2S) and Hydrogen Sulfide (H2S), Phys. Chem. Chem. Phys., 23, 13647-13661 (2021). (PDF)
33. S. J. Goettl, Z. Yang, S. Doddipatla, C. He, M. O. Alves, B. R. L. Galvão, R. I. Kaiser, A Crossed Molecular Beams and Computational Study of the Formation of the Astronomically Elusive Thiosilaformyl Radical (HSiS, X2A'), J. Phys. Chem. Lett., 12, 5979-5986 (2021). (PDF)
34. S. Doddipatla, C. He, S. J. Goettl, R. I. Kaiser, B. R. L. Galvão, T. J. Millar, Nonadiabatic Reaction Dynamics to Silicon Monosulfide (SiS): A Key Molecular Building Block to Sulfur-Rich Interstellar Grains, Sci. Adv., 7, eabg7003 (2021). (PDF)
35. V. S. Krasnoukhov, V. N. Azyazov, A. M. Mebel, S. Doddipatla, Z. Yang, S. Goettl, R. I. Kaiser, Combined Crossed Molecular Beams and Ab Initio Study of the Bimolecular Reaction of Ground State Atomic Silicon (Si; 3P) with Germane (GeH4; X1A1), ChemPhysChem 22, 1497-1504 (2021). (PDF)
36. C. He, S. J. Goettl, Z. Yang, S. Doddipatla, R. I. Kaiser, M. X. Silva, B. R. L. Galvão, Directed Gas-Phase Preparation of the Elusive Phosphinosilylidyne (SiPH2, X2A'') and Cis/Trans Phosphinidenesilyl (HSiPH; X2A') Radicals under Single-Collision Conditions, Phys. Chem. Chem. Phys., 23, 18506 (2021). (PDF)
37. Z. Yang, C. He, S. Goettl, R. I. Kaiser, V. N. Azyazov, A. M. Mebel, Directed Gas-Phase Formation of Aminosilylene (HSiNH2; X1A'): The Simplest Silicon Analogue of an Aminocarbene, under Single-Collision Conditions, JACS, 143, 14227-14234 (2021). (PDF)
38. C. He, S. Doddipatla, Z. Yang, S. J. Goettl, R. I. Kaiser, V. N. Azyazov, A. M. Mebel, T. J. Millar, Gas-Phase Synthesis of Silaformaldehyde (H2SiO) and Hydroxysilylene (HSiOH) in Outflows of Oxygen-rich Asymptotic Giant Branch Stars, Astrophys. J. Lett., 921, L7 (2021). (PDF)
39. A. Rettig, M. Head-Gordon, S. Doddipatla, Z. Yang, R. I. Kaiser, Crossed Beam Experiments and Computational Studies of Pathways to the Preparation of Singlet Ethynylsilylene (HCCSiH; X1A′): The Silacarbene Counterpart of Triplet Propargylene (HCCCH; X3B). J. Phys. Chem Lett. 12, 10768-10776 (2021). (PDF) (Supplemental Information)
40. Z. Yang, C. He, S. J. Goettl, D. Paul, R. I. Kaiser, M. X. Silva, B. R. L. Galvão, Gas-Phase Preparation of Silyl Cyanide (SiH3CN) via a Radical Substitution Mechanism, JACS, 144, 8649-8657 (2022). (PDF)
41. C. He, S. J. Goettl, Z. Yang, R. I. Kaiser, A. A. Nikolayev, V. N. Azyazov, A. M. Mebel, Gas-Phase Preparation of Subvalent Germanium Monoxide (GeO, X1Σ+) via Non-Adiabatic Reaction Dynamics in the Exit Channel, J. Phys. Chem. Lett., 13, 4589-4597 (2022). (PDF)
42. C. He, A. M. Thomas, B. B. Dangi, T. Yang, R. I. Kaiser, H.-C. Lee, B.-J. Sun, A. H. H. Chang, Formation of the Elusive Silylenemethyl Radical (HCSiH2; X2B2) via the Unimolecular Decomposition of Triplet Silaethylene (H2CSiH2; a3A''), J. Phys. Chem. A, 126, 3347-3357 (2022). (PDF)
43. C. He, Y. Luo, S. Doddipatla, Z. Yang, T. J. Millar, R. Sun, R. I. Kaiser, Gas-Phase Formation of Silicon Monoxide via Non-Adiabatic Reaction Dynamics and Its Role as a Building Block of Interstellar Silicates, Phys. Chem. Chem. Phys., 24, 19761-19772 (2022). (PDF)
44. Z. Yang, B.-J. Sun, C. He, J.-Q Li, A. H. H. Chang, R. I. Kaiser, Gas-Phase Preparation of 1-Germavinylidene (H2CGe; X1A1) – the Isovalent Counterpart of Vinylidene (H2CC; X1A1) - via Non-Adiabatic Dynamics through the Elementary Reaction of Ground State Atomic Carbon (C; 3P) with Germane (GeH4; X1A1) , J. Phys. Chem. Lett., 14, 430-436 (2023). (PDF)
45. D. Paul, B.-J. Sun, C. He, Z. Yang, S. J. Goettl, T. Yang, B.-Y. Zhang, A. H. H. Chang, R. I. Kaiser, Competing Si2CH4-H2 and SiCH2-SiH4 Channels in the Bimolecular Reaction of ground-State Atomic Carbon (C(3pj)) with Disilane (Si2H6, X1A1g) under Single Collision Conditions , J. Phys. Chem. A, 127, 1901-1908 (2023). (PDF) (Supplemental Information)
46. Z. Yang, S. Doddipatla, C. He, S. J. Goettl, R. I. Kaiser, A. W. Jasper, A. C. R. Gomes, B. R. L. Galvão, Can third-body stabilisation of bimolecular collision complexes in cold molecular clouds happen? Molecular Physics, e2134832 (2023). (PDF)
47. I. A. Medvedkov, A. A. Nikolayev, S. J. Goettl, Z. Yang, A. M. Mebel, R. I. Kaiser, Experimental and theoretical study of the Sn – O bond formation between atomic tin and molecular oxygen , Phys. Chem. Chem. Phys., 26, 27763–27771 (2024). (PDF) (Supplemental Information) (Data Files)
48. S. J. Goettl, A. Vincent, M. X. Silva, Z. Yang, B. R. L. Galvão, R. Sun, R. I. Kaiser, Gas-Phase Preparation of Silylacetylene (SiH3CCH) Through a Counterintuitive Ethynyl Radical (C2H) Insertion, Sci. Adv., 10, eadq5018 (2024). (PDF)
49. S. Metya, S. J. Goettl, I. A. Medvedkov, M. O. Alves, B. R. L. Galvão, R. I. Kaiser, Gas-Phase Synthesis of Germanium Monosulfide (GeS, X1Σ+) via the Elementary Reaction of Atomic Germanium (Ge, 3P) with Hydrogen Sulfide (H2S, X1A1), J. Phys. Chem. Lett, 16, 4780-4787 (2025). (PDF) (Supplemental Information) (Data Files)
50. S. Metya, I. A. Medvedkov, S. J. Goettl, T. Sattasathuchana, M. X. Silva, B. R. L. Galvão and R. I. Kaiser, One Collision – Two Heteroatoms: Gas-Phase Preparation of Azasilabenzenes, Angew. Chem. Int. Ed., 65, e17656 (2026). (PDF) (Supplemental Information) (Data Files)


